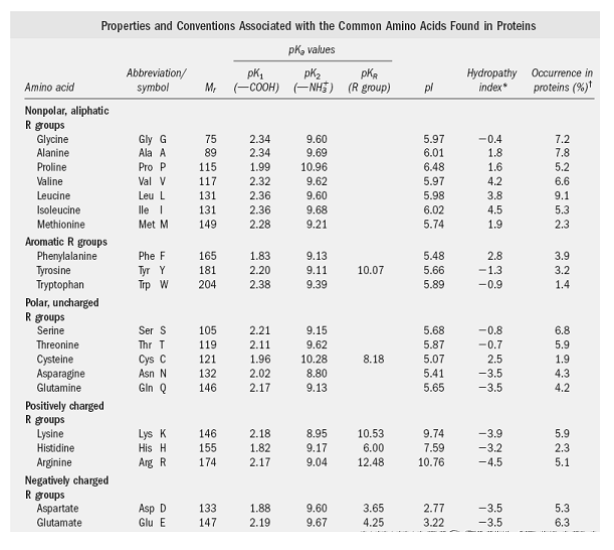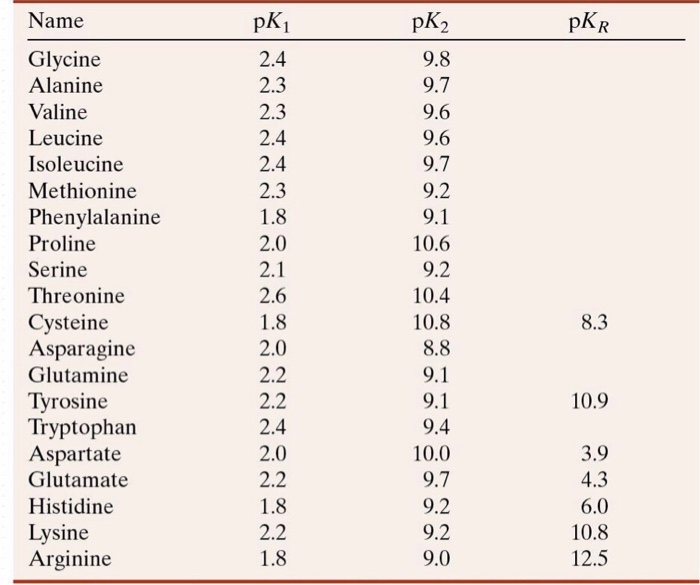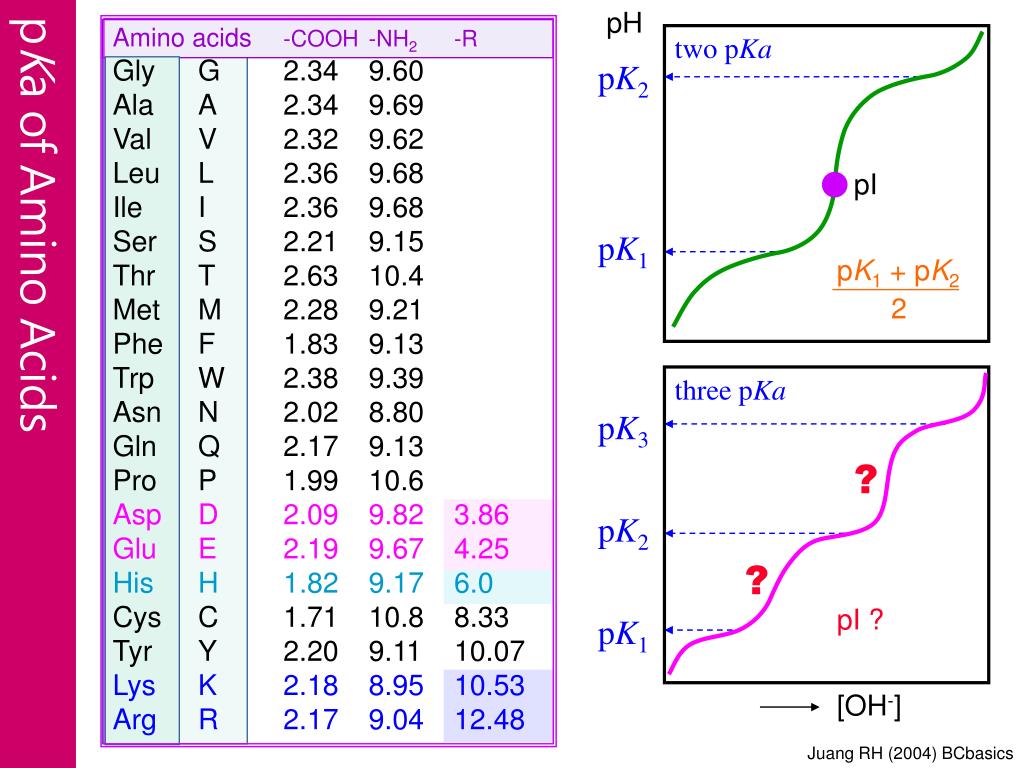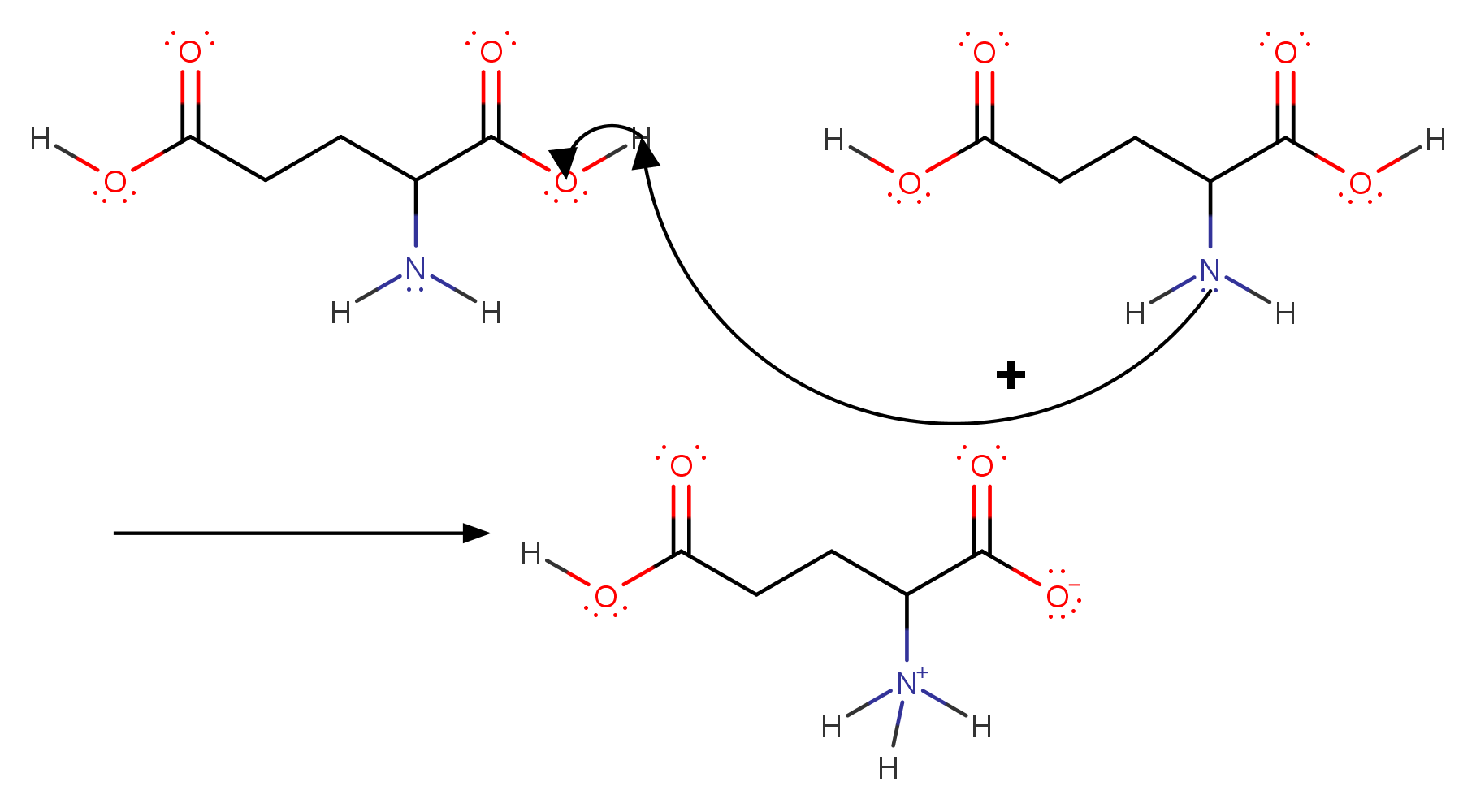
Using the Pka' Values for glutamic acid (pk1= 2.19, pk2= 9.67, pkR= 4.25) indicate the ionic form which predominates at: a) pH 1.0 b)pH 7.0 c) pH13 d) What is the net
BC367 Experiment 1 Identification of an Unknown Amino Acid Introduction As the building blocks of proteins, amino acids play a k

The amino acid methionine has pKa1 = 2.2 and pKa2 = 9.1. If this amino acid is represented by H2L+, what is the major species at pH 6? | Homework.Study.com
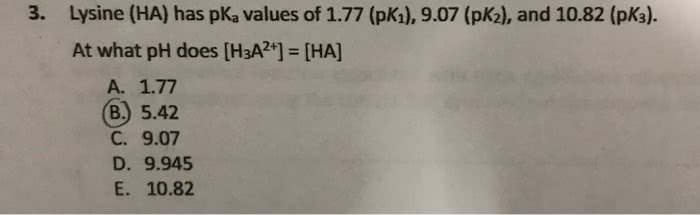
OneClass: 3. Lysine (HA) has pKa values of 1.77 (pKı), 9.07 (pK2), and 10.82 (pK3). At what pH does ...
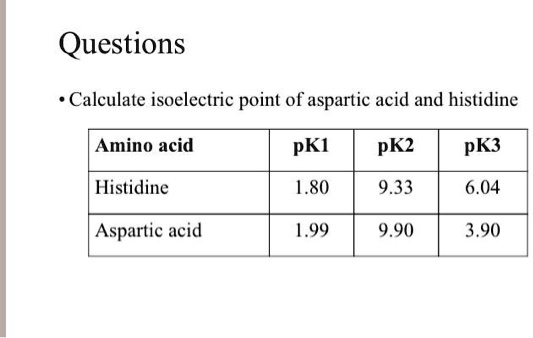
Calculate the isoelectric point of aspartic acid and histidine. Amino acid pKI pK2 pK3 Histidine 1.80 9.33 6.04 Aspartic acid 1.99 9.90 3.90


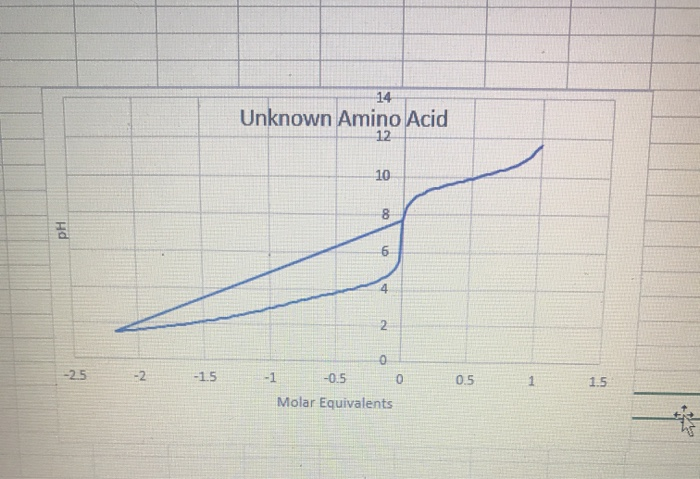


![ANSWERED] Calculate the iso-electric points (pl) for the following - Kunduz ANSWERED] Calculate the iso-electric points (pl) for the following - Kunduz](https://media.kunduz.com/media/sug-question/raw/55454859-1659271618.1019814.jpeg)