
Fast Dispatch Pure Crystal Nacl, Bulk Stock for Water Softening, Ice Management - China Sodium Chloride, Nacl | Made-in-China.com

SOLVED: This stock solution of NaCl (made above in Problem 1) is to be used to prepare a solution containing (10mM NaCl). How much of the 5M NaCl stock solution is used

SOLVED: This stock solution of NaCl (made above in Problem 1) is to be used to make 5mL working solution containing 0.1M NaCl. How much of the 0.1M NaCl stock solution needs
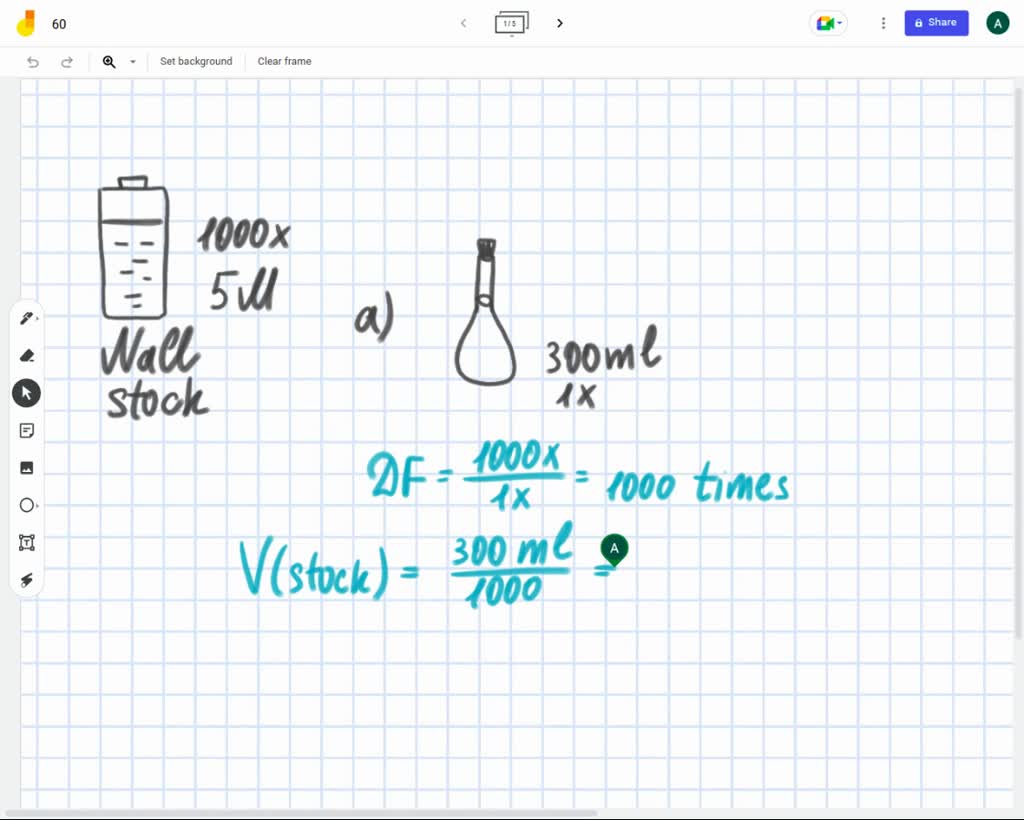
SOLVED: You have a 1000X stock solution of sodium chloride (NaCl). The concentration of NaCl in the stock solution is 5M. a)How would you make 300mL of a 1X solution? b)What is

You are preparing 500 mL of Denaturation Buffer containing 1.5 M NaCl and 0.5 M NaOH. Your stock solutions are 5 M NaCl and 6 M NaOH. How would you prepare the

Describe how you would prepare 10 mL each of 2 M, 1.5 M, and 1 M NaCl solutions from a 2 M stock NaCl solution. Describe your actions exactly what you would
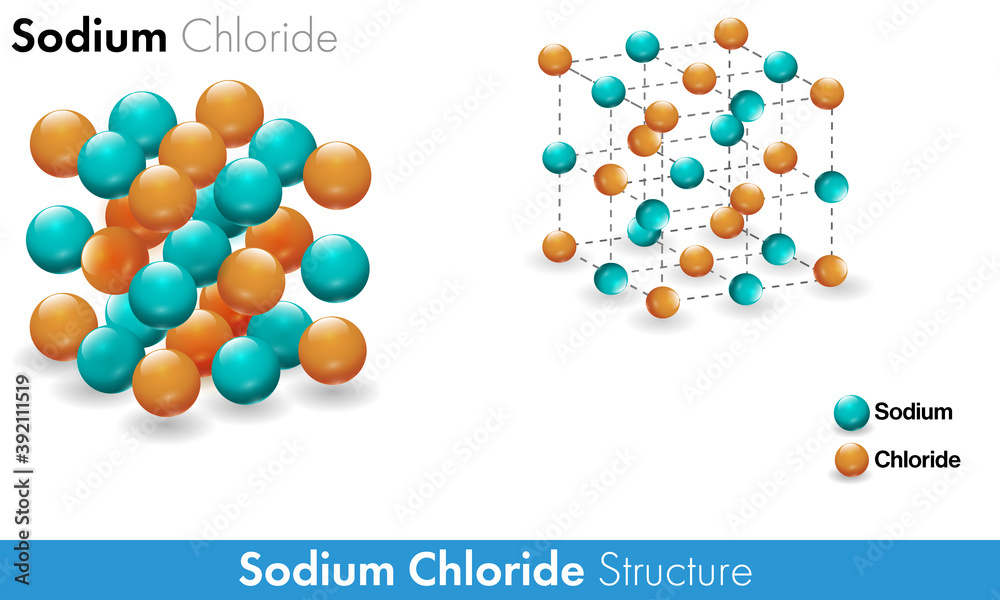


![ANSWERED] A 10 M concentrated stock solution of NaCl was used to - Kunduz ANSWERED] A 10 M concentrated stock solution of NaCl was used to - Kunduz](https://media.kunduz.com/media/sug-question/raw/49152426-1658686694.5703318.jpeg)



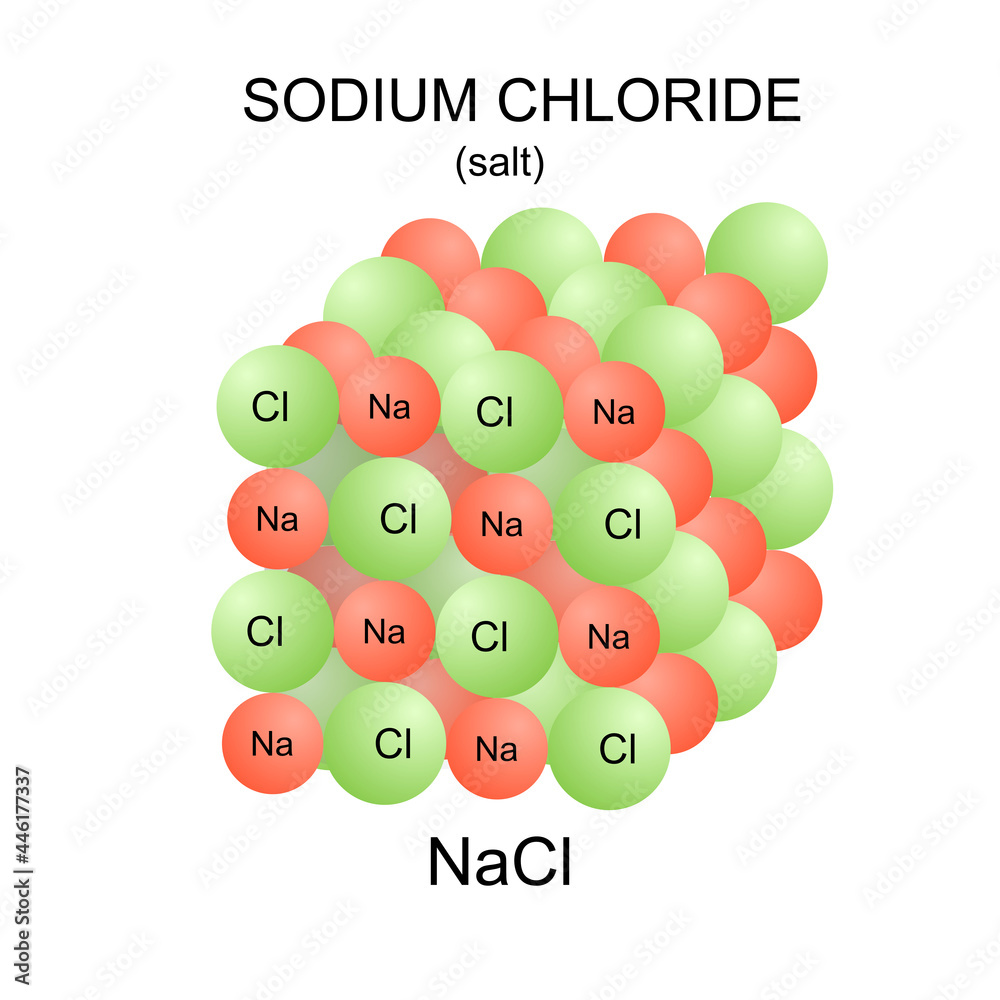


![Sodium chloride, NaCl structure chemistry,... - Stock Illustration [98084788] - PIXTA Sodium chloride, NaCl structure chemistry,... - Stock Illustration [98084788] - PIXTA](https://en.pimg.jp/098/084/788/1/98084788.jpg)
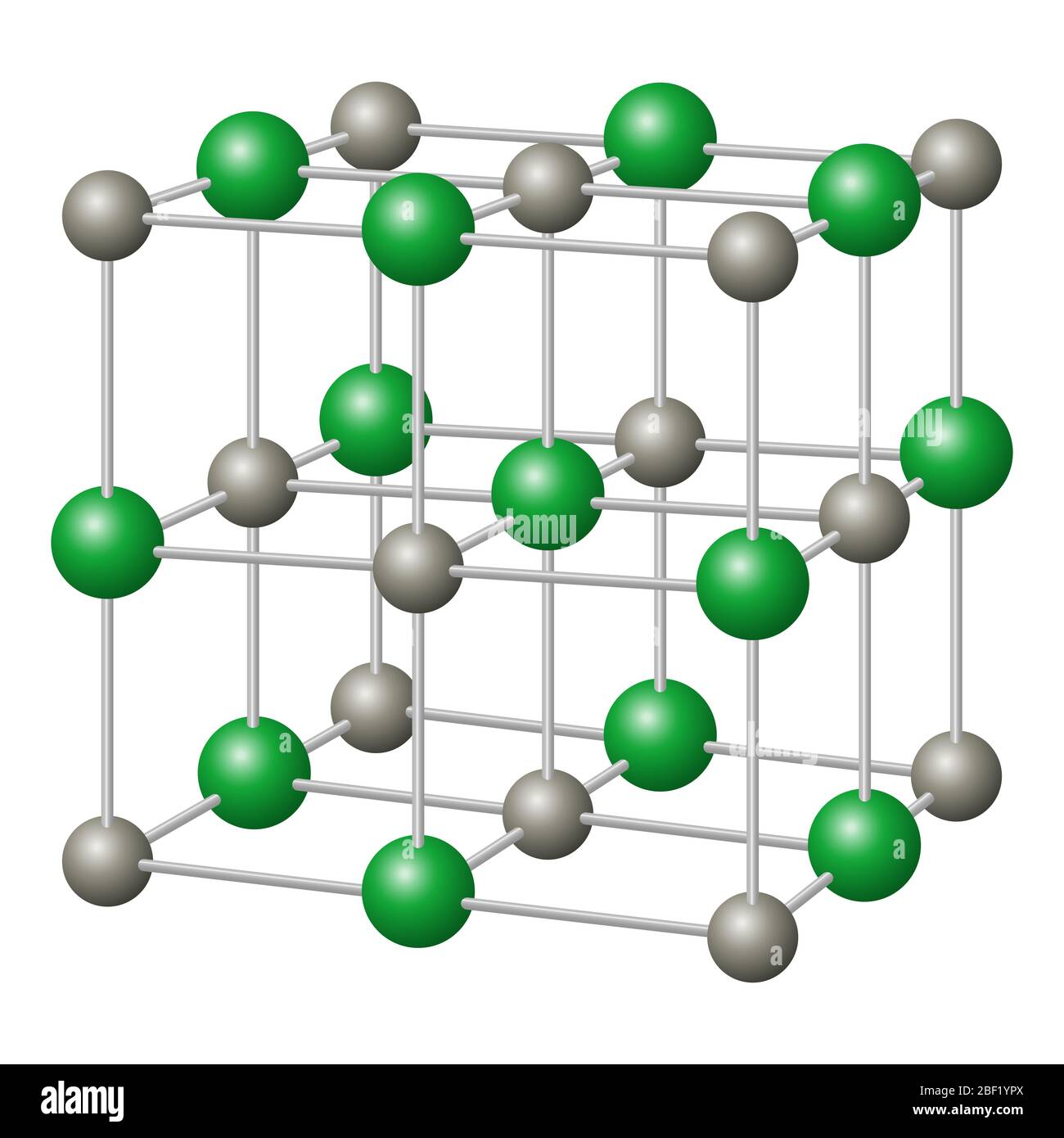


![Sodium chloride, NaCl crystal structure with... - Stock Illustration [64444522] - PIXTA Sodium chloride, NaCl crystal structure with... - Stock Illustration [64444522] - PIXTA](https://en.pimg.jp/064/444/522/1/64444522.jpg)



