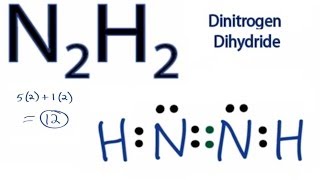OneClass: b. NF3 e SCl2 C. BraN 2. Determine whether each molecule is polar or nonpolar, and justify ...

N2H2 Lewis Structure (Dinitrogen Dihydride) | N2H2 Lewis Structure (Dinitrogen Dihydride) Today in this video we help you determine the Lewis Structure of Dinitrogen Dihydride. The molecule has a... | By Geometry
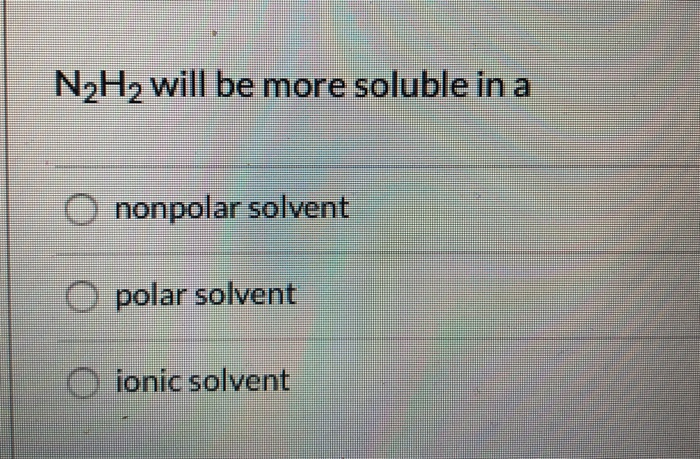
N2H2 is polar molecule with london dispersion forces,dipole dipole forces and H bonding .True or false?How?
Why does N, N Dimethylhydrazine have 'hydrazine' when it has N2H2, not N2H4 (hydrazine)? Why isn't it called 'Dimethyldiimide'? - Quora