
An Efficient Synthesis of Nα-Protected Amino and Peptide Acid Aryl Amides via Iodine-Mediated Oxidative Acylation of Nα-Protected Amino and Peptide Thioacids

Synthesis of C-Propargylic Esters of N-Protected Amino Acids and Peptides | The Journal of Organic Chemistry
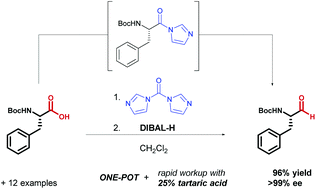
A rapid and efficient one-pot method for the reduction of N-protected α-amino acids to chiral α-amino aldehydes using CDI/DIBAL-H - Organic & Biomolecular Chemistry (RSC Publishing)

Solventless Synthesis of N-Protected Amino Acids in a Ball Mill | ACS Sustainable Chemistry & Engineering

Scheme 1. The stereoselective transformation of N-protected α-amino... | Download Scientific Diagram
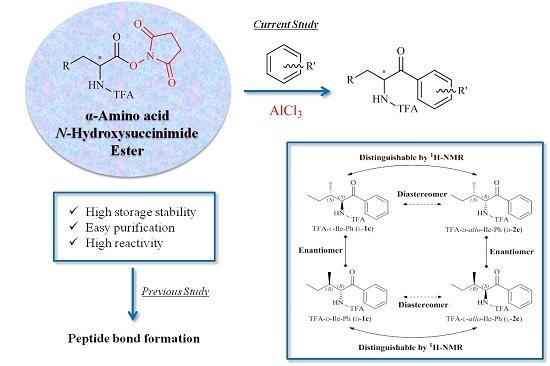
Molecules | Free Full-Text | Synthesis of Chiral TFA-Protected α-Amino Aryl-Ketone Derivatives with Friedel–Crafts Acylation of α-Amino Acid N-Hydroxysuccinimide Ester

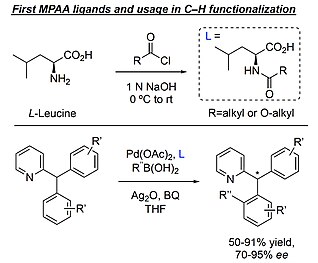
Catalytic Behavior of Mono‐N‐Protected Amino‐Acid Ligands in Ligand‐Accelerated C−H Activation by Palladium(II) - Salazar - 2020 - Angewandte Chemie International Edition - Wiley Online Library

The mechanism of palladium(II)-mediated C–H cleavage with mono-N-protected amino acid (MPAA) ligands: origins of rate acceleration




![Dnp-Amino Acids [N-Protected Amino Acids] | TCI AMERICA Dnp-Amino Acids [N-Protected Amino Acids] | TCI AMERICA](https://www.tcichemicals.com/medias/structure-890-D1033.jpg-Tci-300?context=bWFzdGVyfHJvb3R8MTM2NTd8aW1hZ2UvanBlZ3xoYWQvaDM5LzkzNjU2NDQ2MDc1MTgvc3RydWN0dXJlLTg5MC9EMTAzMy5qcGdfVGNpLTMwMHw5N2RkM2YzMDY2ZDBmYTRkZjY3MWU2MmRmMDA0ZDQ3NTZmMGExMWZhOTc4NmRlMTU1MTdkMzUyZGE2MWExMTEy)
![Ac-Amino Acids [N-Protected Amino Acids] | TCI AMERICA Ac-Amino Acids [N-Protected Amino Acids] | TCI AMERICA](https://www.tcichemicals.com/medias/A0873.jpg?context=bWFzdGVyfHJvb3R8MzEzNjB8aW1hZ2UvanBlZ3xoNTkvaGViLzg5MjgyNjAyNTk4NzAvQTA4NzMuanBnfDg1OTZkOTU4NmI0NzhiMzE2NmFlYjQ5M2RkODkxZjFiOGQ5ZmRkYWZkNDUzMGMzZGZlYTZkNjMxZWM4YjM4YjU)
![Cbz-Amino Acids [N-Protected Amino Acids] | TCI AMERICA Cbz-Amino Acids [N-Protected Amino Acids] | TCI AMERICA](https://www.tcichemicals.com/medias/C0573.jpg?context=bWFzdGVyfHJvb3R8MzI4MTV8aW1hZ2UvanBlZ3xoNzgvaDliLzg5MjkzMjkxNTIwMzAvQzA1NzMuanBnfGJlZTI2MDg0NDFkOGQ5ZWRkZWVkYzgzMzhkOTMzOTE2YjFkMjY2ZDliYTE2ZjBhYWY1ZDRmYzUxM2IwZTY4OTI)





