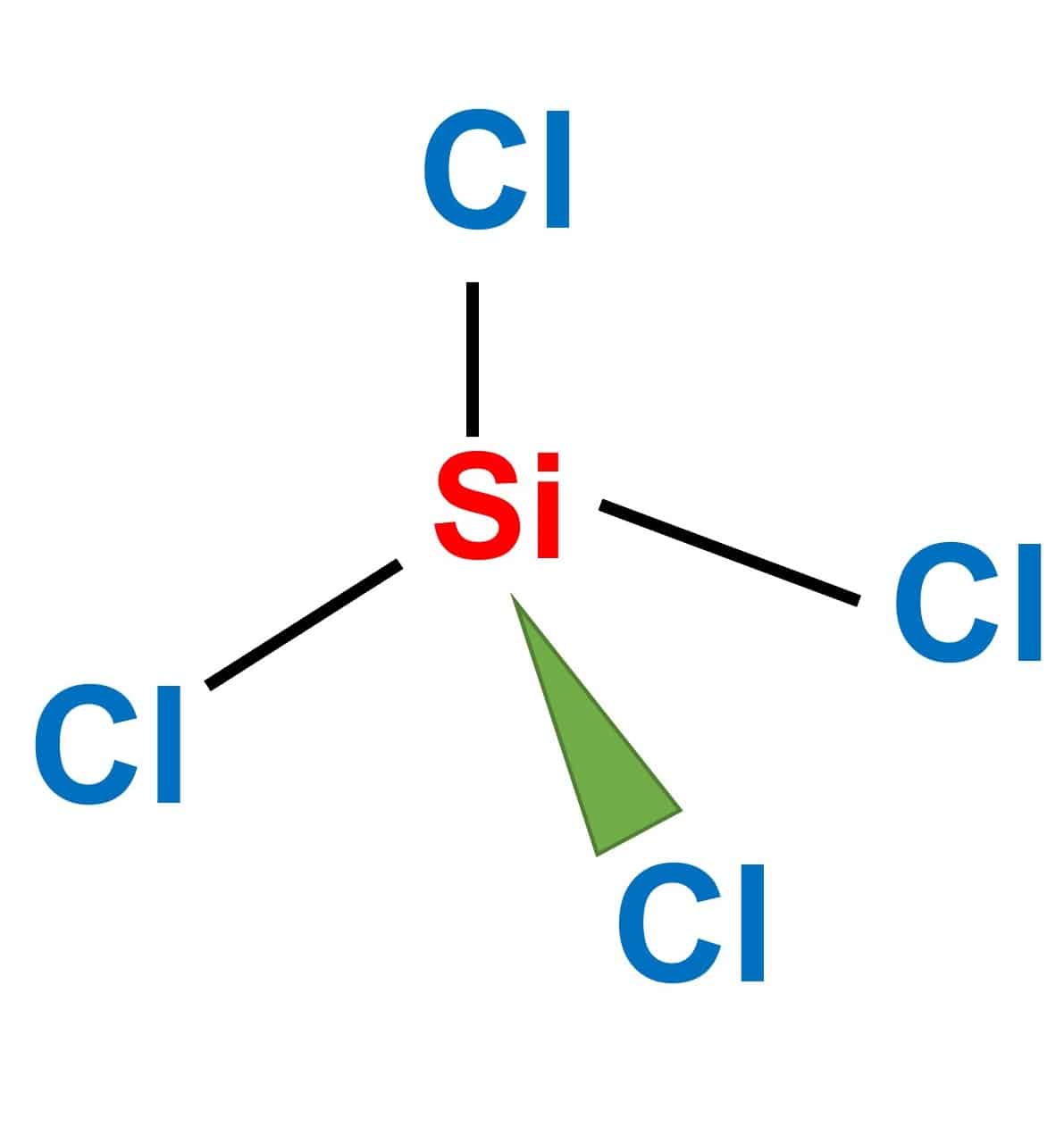
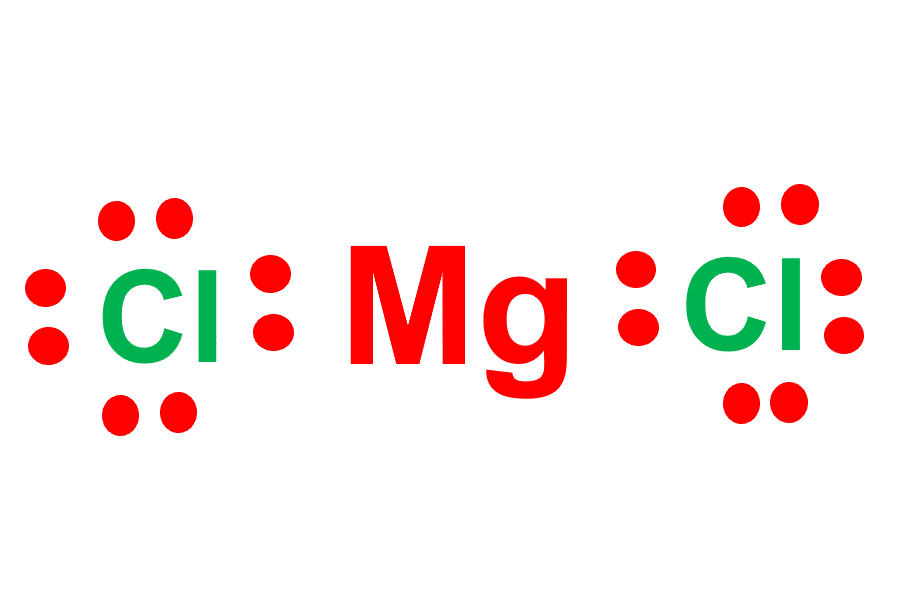Chemistry 1110 Covalent Properties I. Assign electronegativity values to the following elements: N I S P H B O F Br C II. Tell

Elucidating Ionic Correlations Beyond Simple Charge Alternation in Molten MgCl2–KCl Mixtures | The Journal of Physical Chemistry Letters
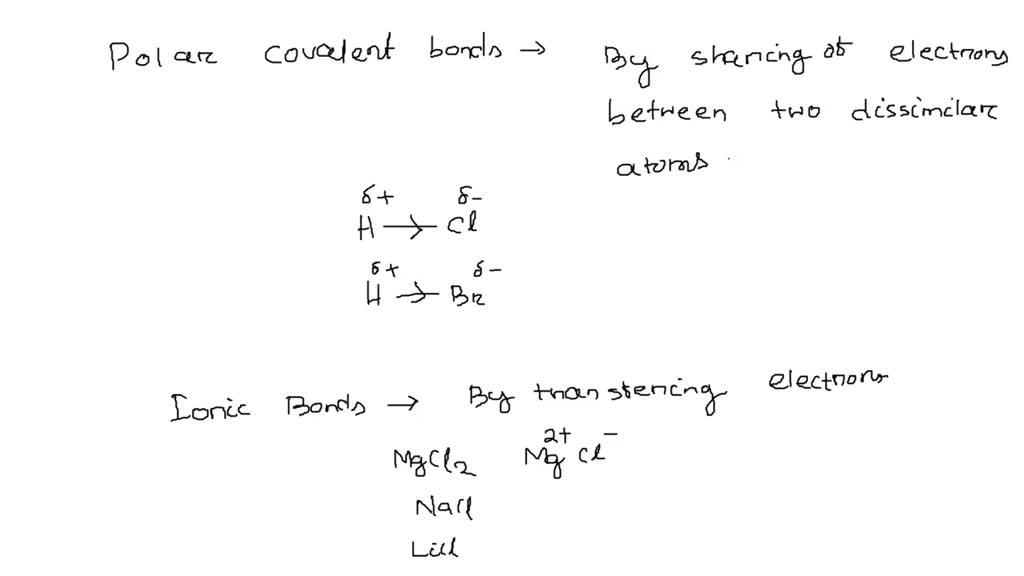
SOLVED: Which of the following substances contain polar covalent bonds? SELECT ALL THAT APPLY- O2- MgCl2- HCl- NaCl- N2- HBr- F2- LiCl

SCN- Formal charge, How to calculate it with images? | Covalent bonding, Chemical bond, Interesting information
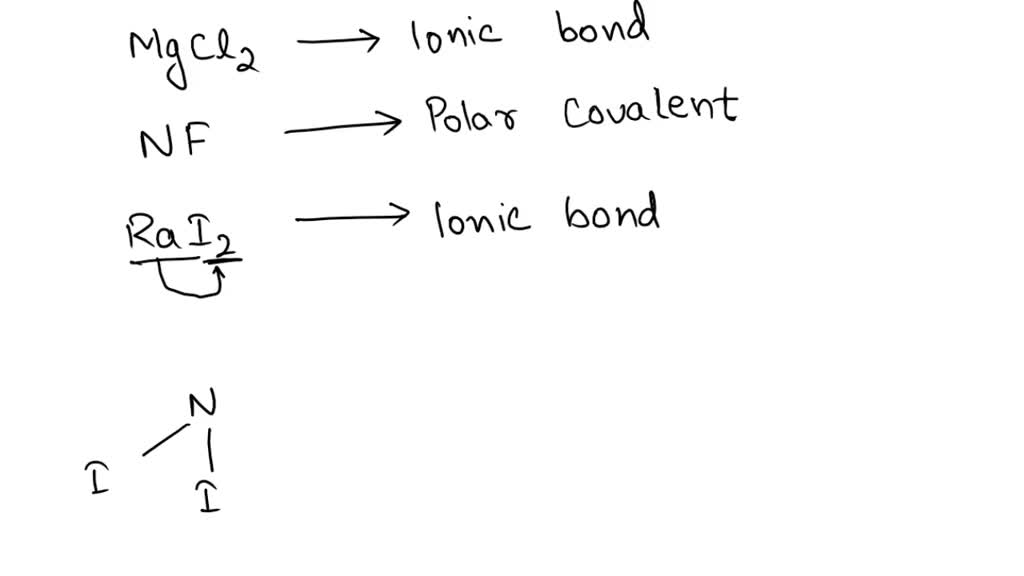
SOLVED: Determine the type of bond (ionic bond, polar covalent bond, or nonpolar covalent bond) present in each of the following elements below: a. CICI - nonpolar covalent bond b. HF -

Which of the following substances have permanent dipole-dipole forces? GeH4; molecular MgCl2; PI3; F2O | Homework.Study.com