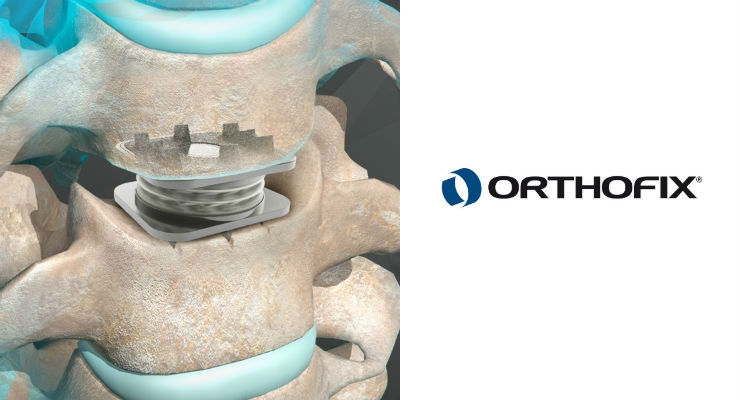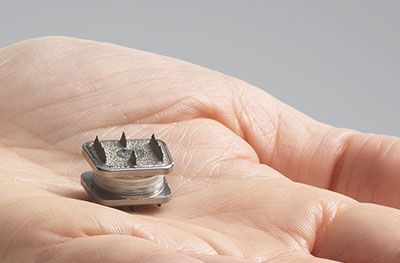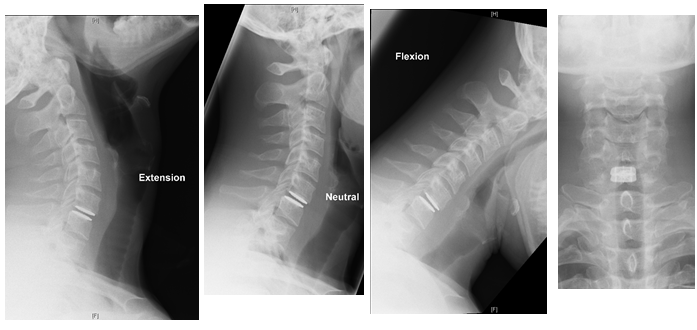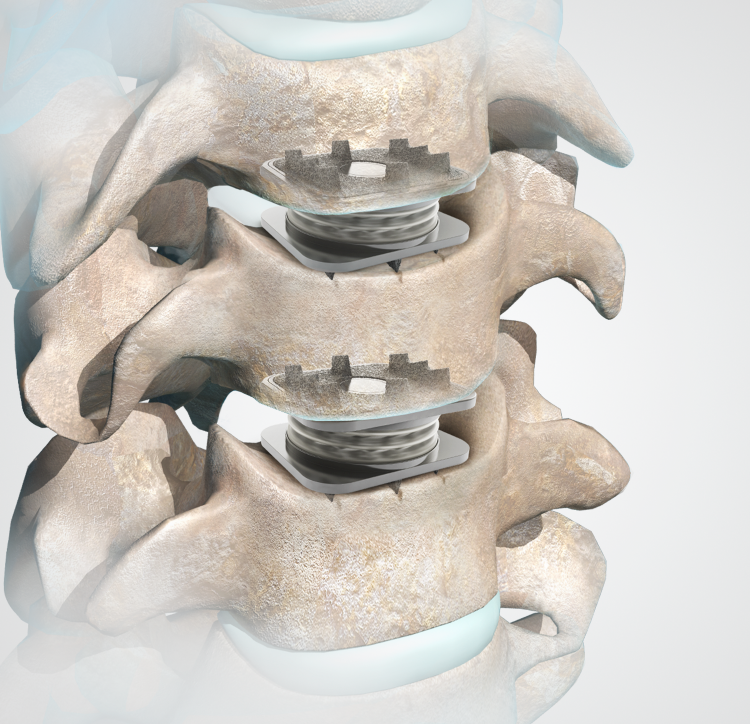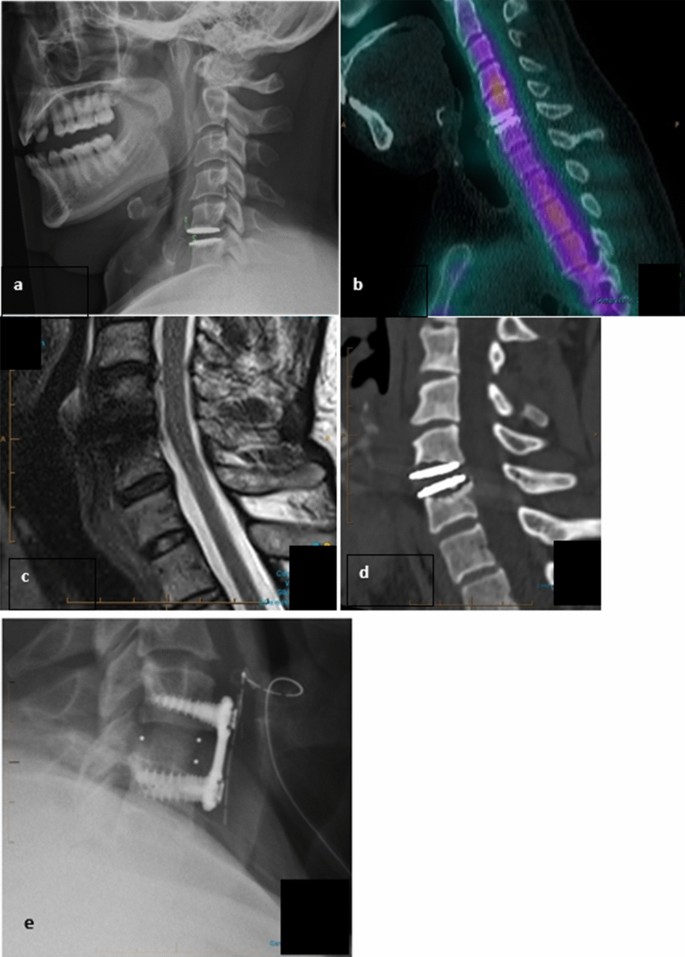
Midterm osteolysis-induced aseptic failure of the M6-C™ cervical total disc replacement secondary to polyethylene wear debris | European Spine Journal
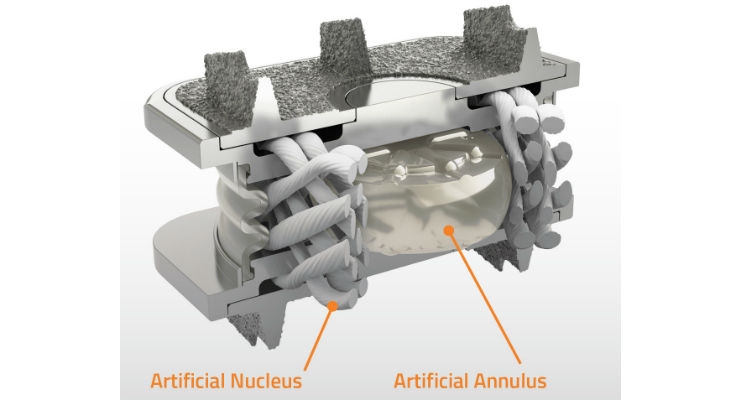
FDA Approves Orthofix's M6-C Artificial Cervical Disc For Cervical Disc Degeneration Treatment | Orthopedic Design Technology