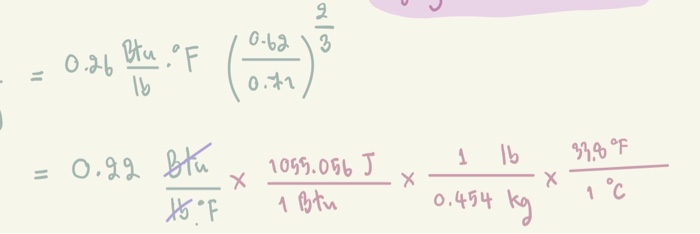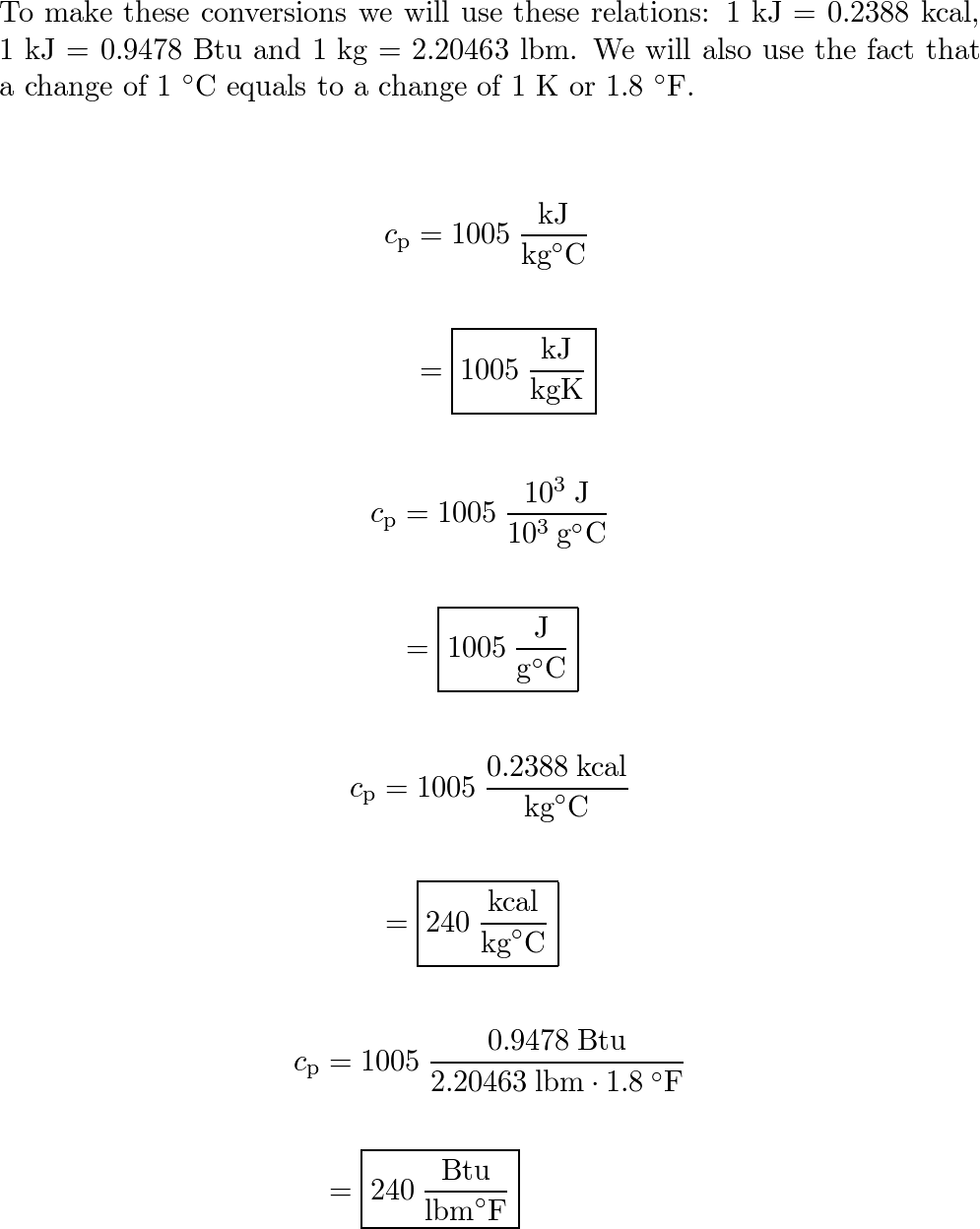SOLVED: Convert the following from English units to SI units: C = 0.25 Btu /(lb·F) to W·s/(kg·C) C = 0.25 Btu/(lb·F) to kJ/(kg·C) k = 0.0263 Btu /(h·ft·F) to W/(m·C) = 0.072 lb/(ft·h) to

Air Has A Specific Heat Of 1 Kjkg K If 2 Btu Of Energy Is Added To 100 G Of Air - MATH001 | Course Hero

SOLVED: The constant-pressure specific heat of air at 175°C is 1.020 kJ /kg°C. Express this value in kJ/kgK, J/g°C, kcal/kg°C, and Btu/lbm°F. kJ/kgK J/g°C kcal/kg°C Btu/lbm°F