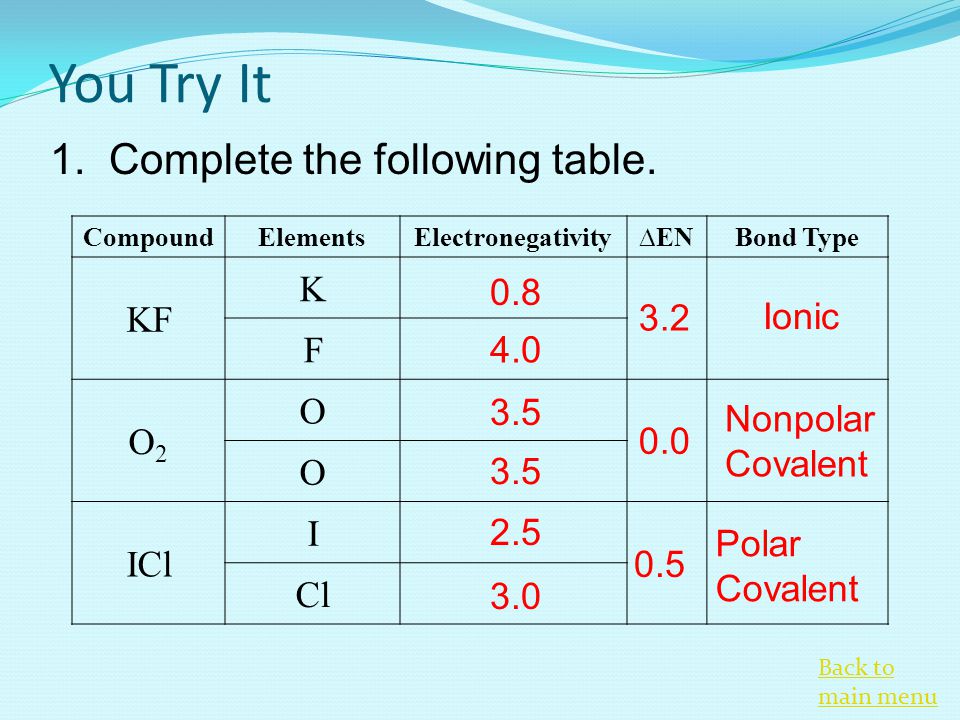
SOLVED: Identify whether the following bonds are: nonpolar covalent; polar covalent; or ionic: K-F N-C H-O C-O

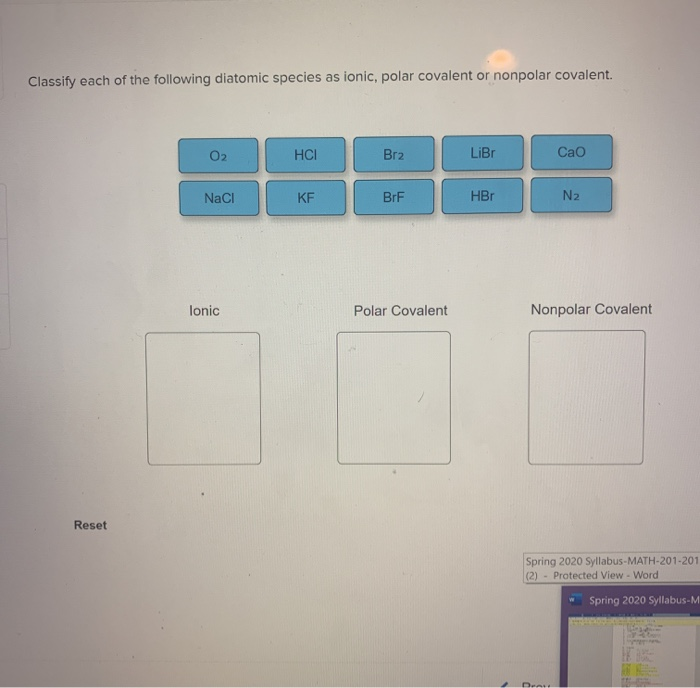
Classify each of the following diatomic species as ionic, polar covalent or nonpolar covalent. | Homework.Study.com
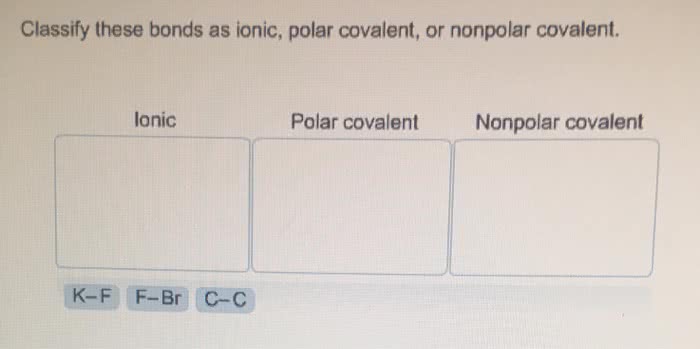
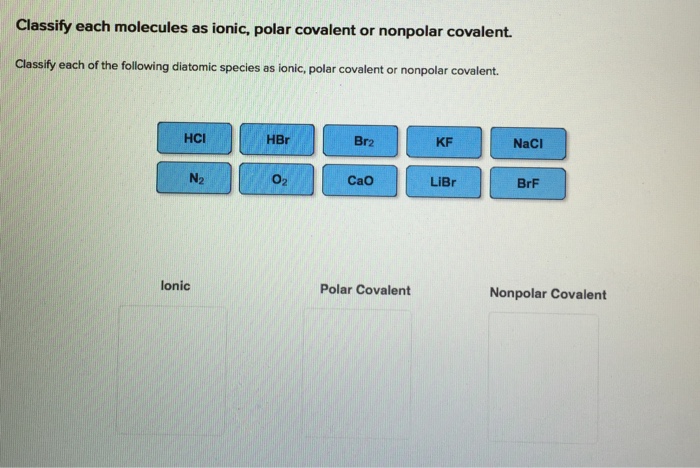
OneClass: Classify these bonds as ionic, polar covalent, or nonpolar covalent. lonic Polar covalent N...

HYDRANAL™ - LipoSolver MH, Reagent for volumetric one-component KF titration in non-polar substances, fats
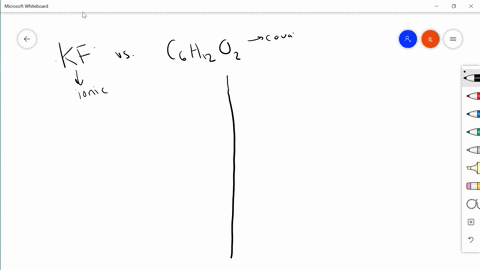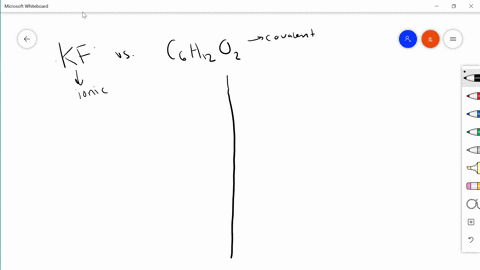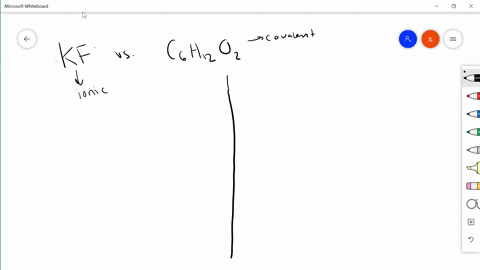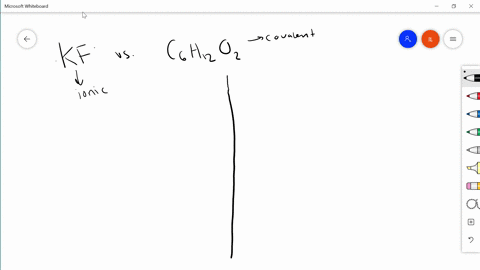
SOLVED:Differentiate between what happens when the following are added to water. a. polar solute versus nonpolar solute b. KF versus C6 H12 O6 c. RbCl versus AgCl d. HNOversus CO