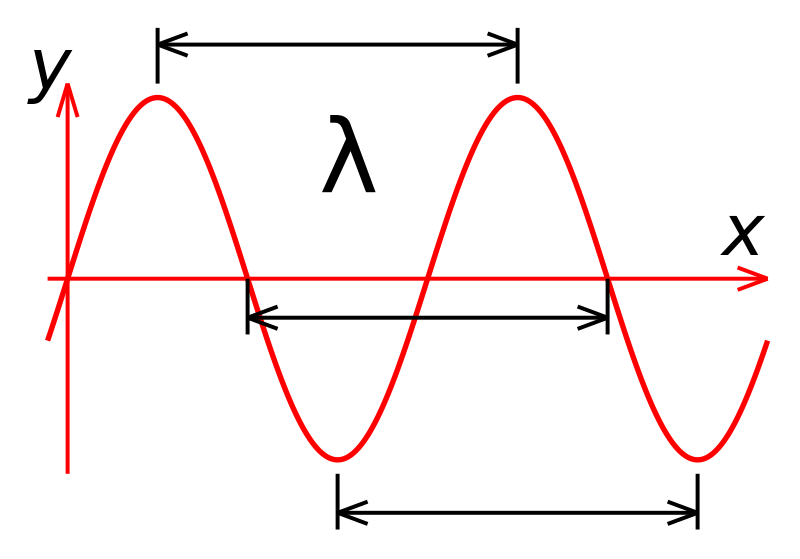
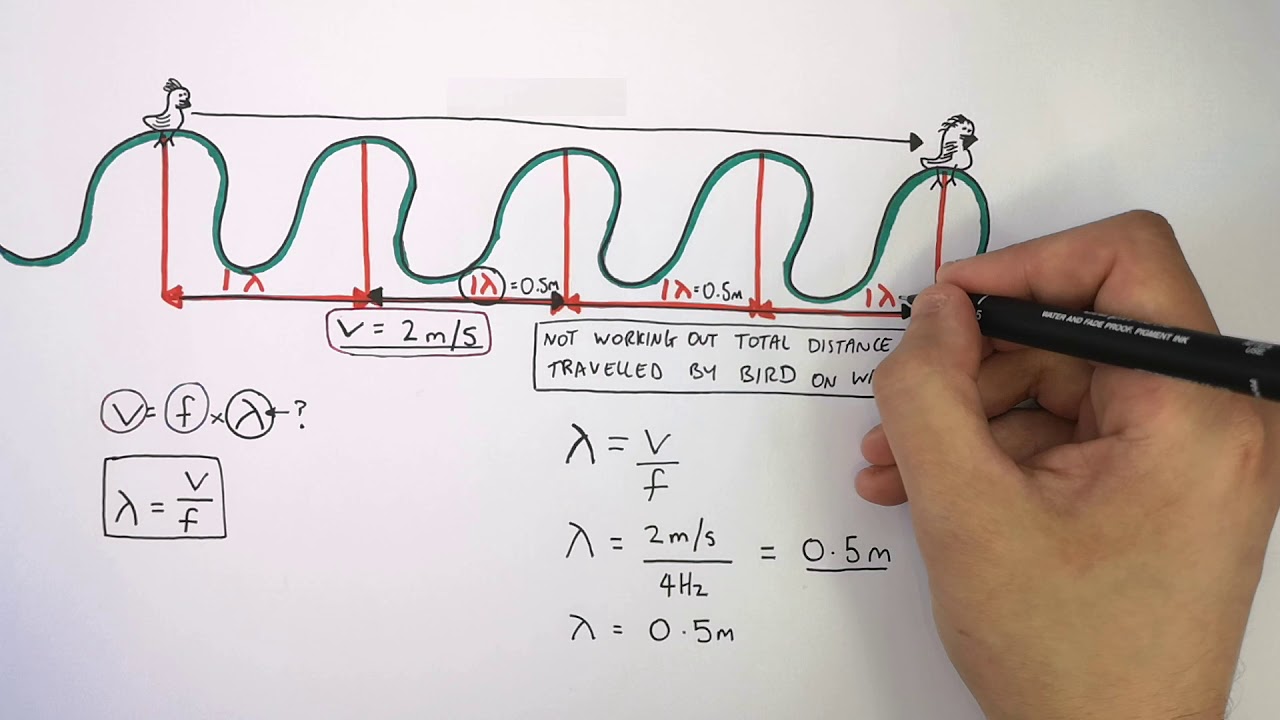
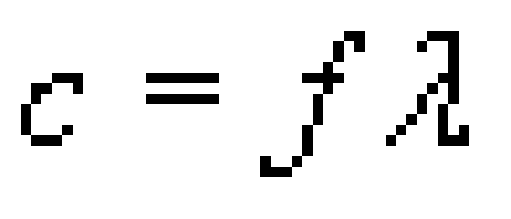Hydrogen (1H^1) , Deuterium (1H^2) , singly ionised Helium (2He^4)^ + and double ionised lithium (3Li^6)^++ all have one electron around the nucleus. Consider an electron transition from n = 2 to
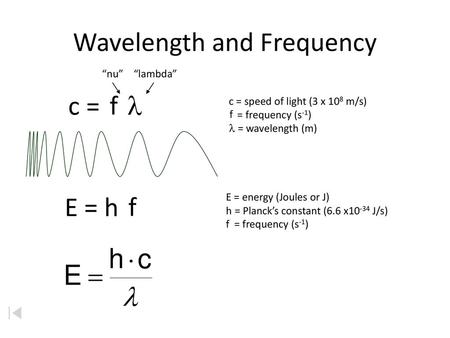
Wavelength and Frequency E = h c = c = speed of light (3 x 10 8 m/s) = frequency (s -1 ) = wavelength (m) E = energy (Joules or J) h = Planck's constant. - ppt download