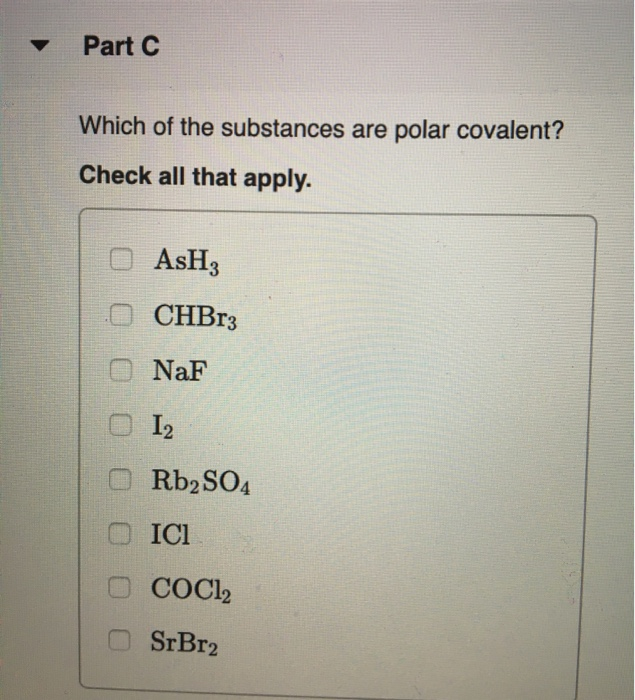
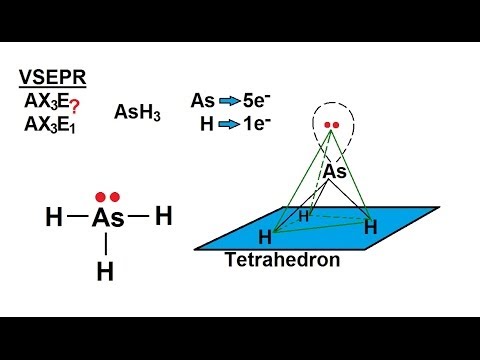
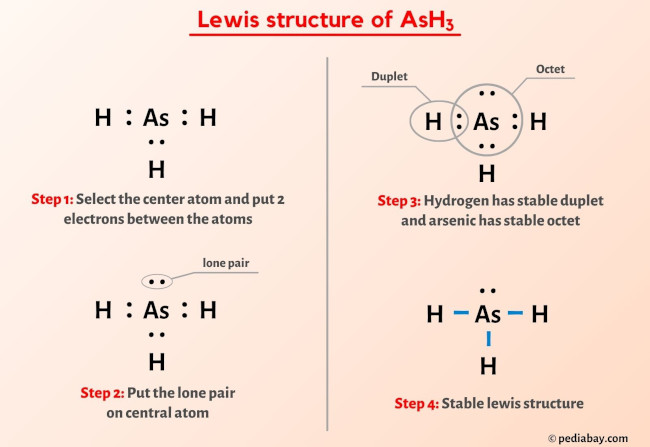
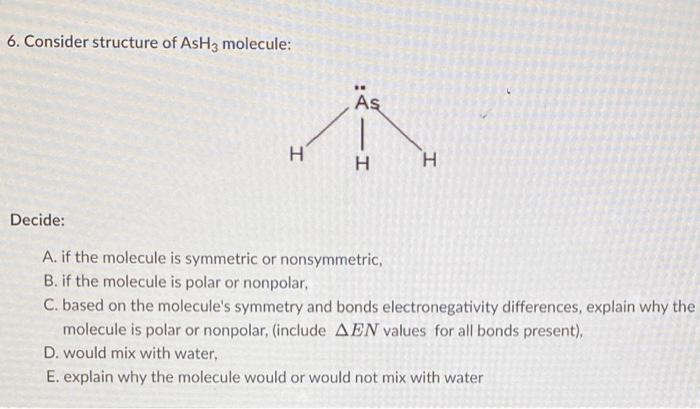
SOLVED: What is the molecular geometry and molecular polarity for AsH3? Linear nonpolar Linear ionic Bent nonpolar Bent ionic Bent polar Trigonal planar polar Tetrahedral ionic Trigonal pyramidal nonpolar Trigonal pyramidal polar

SOLVED: An AX formula corresponds to what degree of hybridization? How many hybridized orbitals are nonbonding in each of the following? H2O BF3 NH3 SF4 How many sigma bonds are in each